Children’s Nebraska’s goal is to provide the highest level of care for the community’s children, as well as support the primary care providers who care for them. We want to share with you information on our process for testing pediatric patients for COVID-19 and information on screening for Multisystem Inflammatory Syndrome in Children (MIS-C).
Informational COVID-19 resources to share with families:
Find helpful COVID-19 resources from our Max The Vax and Fruitful COVID-19 Vaccine toolkits at https://maxthevaxne.org/provider-toolkit/ or join more than 180 physicians and providers from across Nebraska who have signed a Statement of Support for FDA-Approved COVID-19 Vaccinations for kids five and older at https://www.neprovidersforthevax.org.
COVID-19 Testing Process
If you are affiliated with a health system with a lab that is processing COVID-19 testing, please have your patient tested through your affiliated system.
If you are not affiliated with a health system with a lab that is processing COVID-19 testing, and you have a patient that needs to be tested, please follow the steps below:
- Have the legal guardian call Children’s Access Center at 402-955-4066 to complete the registration process and let them know the child is coming to the Respiratory Assessment Center (RAC) with a prescription.
- Send a paper prescription with the patient.
- The RAC is located on the east side of Children’s, and can be accessed from 84th Street between West Dodge Road and Indian Hills Drive. Families should turn into the entrance by the large white tent and follow the directional signage. They will then be greeted and screened by Children’s staff. Please note: The patient should be accompanied by an adult who has NOT tested positive for COVID-19.
If you have a symptomatic patient who has not been physically seen by you, please follow these steps:
- Direct the parent to call Children’s COVID-19 Hotline at 402-955-3200.
- A virtual visit will be scheduled. If it’s deemed appropriate, the patient will be sent to the RAC for a physical assessment and test. All documentation and test results will be available for your review via Physician Connect.
Please note: You will be responsible for contacting your patient families with results and reviewing self-isolation guidance based on test results.
COVID-19 Treatments
Approved or Emergency Use Authorized (EUA) COVID-19 treatments continue to change based on current variants. COVID-19 monoclonal antibodies have been used to treat and prevent some COVID-19 variants. Current variants in the US are resistant to all available COVID-19 monoclonal antibodies and therefore there is no FDA authorized monoclonal antibody to treat or prevent COVID-19.
Available COVID-19 Treatments:
1. Paxlovid (nirmatrelvir and ritonavir) Capsules
Indication: FDA approved for the treatment of adults and EUA for pediatric patients (12 years of age and older weighing at least 40 kg) with a diagnosis of mild-to-moderate COVID-19, and who are at high risk for progression to severe COVID-19, including hospitalization or death.
Availability: Local outpatient pharmacies.
Limitations: Many drug-drug interactions. Healthcare providers need to verify no drug-drug interactions prior to prescribing Liverpool COVID-19 Interactions (covid19-druginteractions.org), review age/weight limitations, and renally adjust dosing if necessary.
2.Veklury (remdesivir) intravenous infusion
Indication: FDA approved for treatment of COVID-19 in adults and pediatric patients weighing at least 1.5kg who are: 1) hospitalized or 2) non-hospitalized and have mild to moderate COVID-19, and are at high risk for progression to severe COVID-19, including hospitalization or death. Remdesivir is administered by intravenous infusion daily for 3 consecutive days if given outpatient. Inpatient treatment may be longer.
Availability: Select hospitals and infusion centers including Children’s Nebraska. For outpatient use at Children’s Nebraska follow the following steps.
Limitations: Expense, insurance deductibles may need to be met and prior authorization may be needed. Multiple days of IV treatment.
Screening for Multisystem Inflammatory Syndrome in Children
Although MIS-C is a rare occurrence, quick diagnosis is important in expediting positive outcomes. Children’s hospitalist team has developed a MIS-C screening algorithm.
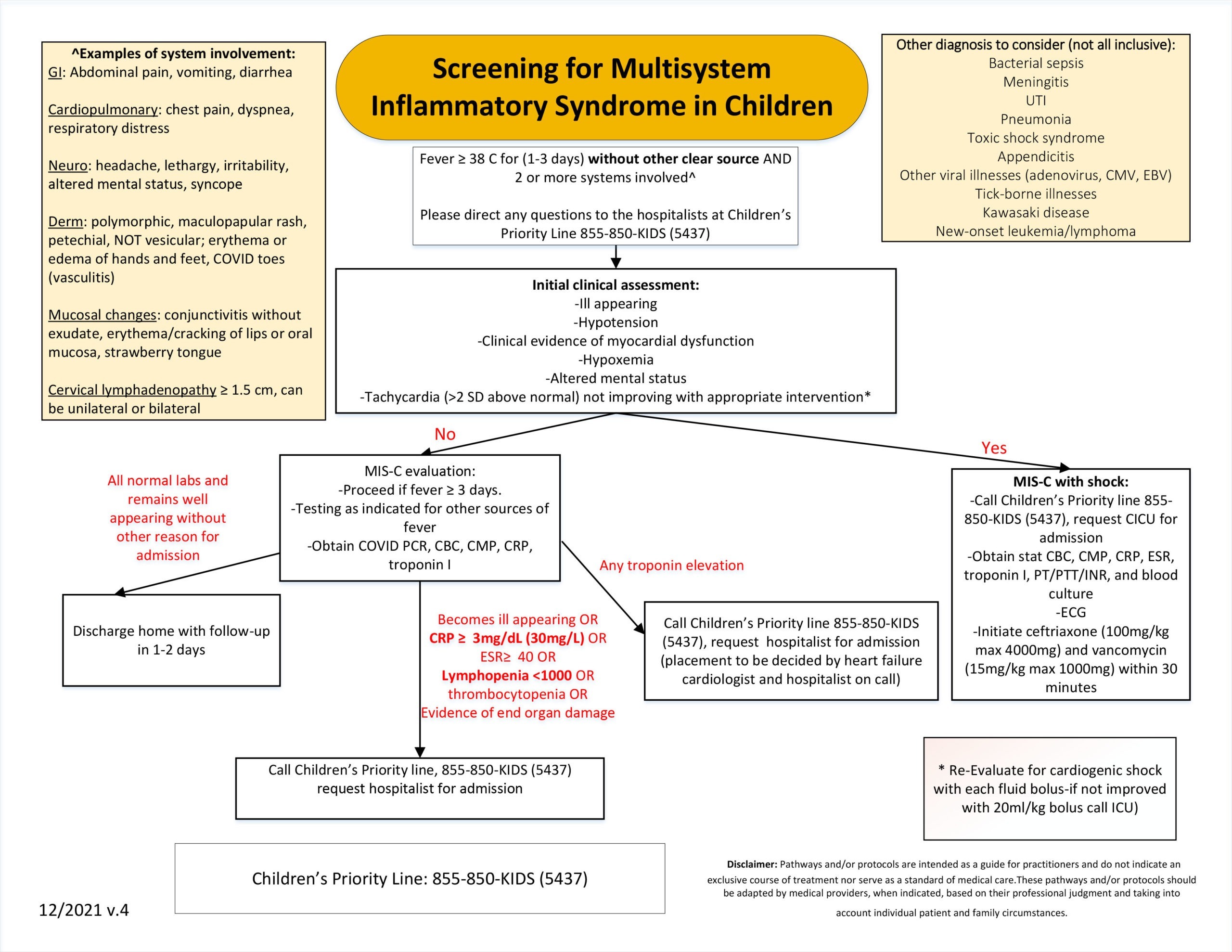
CHILDREN’S PRIORITY LINE: 855-850-KIDS (5437)
Disclaimer: Pathways and/or protocols are intended as a guide for practitioners and do not indicate an exclusive course of treatment nor serve as a standard of medical care. These pathways and/or protocols should be adapted by medical providers, when indicated, based on their professional judgment and taking into account individual patient and family circumstances.
Download PDF of MIS-C screening algorithm.
Please review these guidelines and if you have any non-emergent questions, contact Dr. Aleisha Nabower at [email protected].
For an immediate consult, please call Children’s Physicians Priority Line at 855-850-5437.
Acute COVID-19 Infection vs. MIS-C

Educational Video